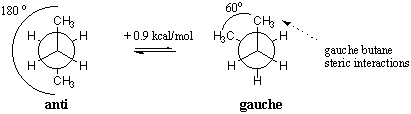 The fluorine gauche effect in 1,2‐difluoroethane and the related anomeric effect.
The fluorine gauche effect in 1,2‐difluoroethane and the related anomeric effect.
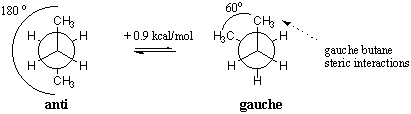 This effect is present in 1,2-difluoroethane (H 2 FCCFH 2) for which the gauche conformation is more stable by 2.4 to 3.4 kJ/mole in the gas phase.
This effect is present in 1,2-difluoroethane (H 2 FCCFH 2) for which the gauche conformation is more stable by 2.4 to 3.4 kJ/mole in the gas phase.
Source : Gauche Effect in 1,2-Difluoroethane. 1,2-difluoroethane. 1,2-Difluoroethane is widely recognised to adopt a lower energy gauche rather than anti conformation; this gauche effect has its origin in hyperconjugation; however, surprisingly the 1 J CF coupling constant is not influenced by hyperconjugation; instead, its magnitude changes with the overall molecular dipole. Effect of Orbital Interactions between Vicinal Bonds and between Hydroxy Groups on the Conformational Stabilities of 1,2-Ethanediol and 2,3-Butanediols Nobuyuki Hayashi , Tomomi Ujihara , … It is well established that vicinal fluorines (RCHF-CHFR) prefer to adopt a gauche rather than an anti conformation when placed along aliphatic chains.This has been particularly recognised for 1,2-difluoroethane and extends to 2,3-difluorobutane and longer alkyl chains. The gauche effect is very sensitive to solvent effects, due to the large difference in polarity between the two conformers.For example, 2,3-dinitro-2,3-dimetylbutane, which in the solid state exists only in the gauche conformation, prefers the gauche conformer in benzene solution by a ratio of 79:21, but in carbon tetrachloride it prefers the anti conformer by a ratio of 58:42. Another example is 1,2-dimethoxyethane. The relative stability of the gauche-conformation of 1,2-difluoroethane is found to be a consequence of the nodal structure of the singly occupied orbital in the CFH 2 radical. There are two main explanations for the gauche effect: hyperconjugation and bent … Another phenomenon that Fleming touches on which he uses the same explanation for is the preference for the gauche conformation in 1,2-difluoroethane. (2)School of Chemistry, University of Manchester , Oxford Road, Manchester M13 9PL, Great Britain. The Gauche effect characterizes any gauche rotamer which is actually more stable than the anti rotamer. (2)School of Chemistry, University of Manchester , Oxford Road, Manchester M13 9PL, Great Britain. According to NMR three‐bond spin‐spin coupling constants, the gauche population increases when fluorine is replaced by a hydroxyl group (except for 2‐fluoroethanol relative to 1,2‐difluoroethane), but this is not primarily due to intramolecular hydrogen bond. This calculates to a postmortem CB/PB ratio of 1.47, compared to an average of 1.80 for similar cases at the SDCMEO. Gauche Effect in 1,2-Difluoroethane. It is also shown that the nodal structure of the singly occupied orbitals in the CFH biradical can explain the stability of the cis conformation of 1,2-difluoroethene. Another example is 1,2-dimethoxyethane. Another example is 1,2-dimethoxyethane. The Gauche effect characterizes any gauche rotamer which is actually more stable than the anti rotamer . Gauche effect . Hyperconjugation, Bent Bonds, Steric Repulsion Hyperconjugation, Bent Bonds, Steric Repulsion Goodman, Lionel
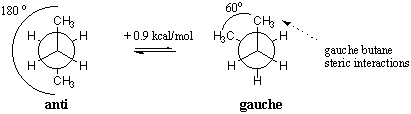 The fluorine gauche effect in 1,2‐difluoroethane and the related anomeric effect.
The fluorine gauche effect in 1,2‐difluoroethane and the related anomeric effect.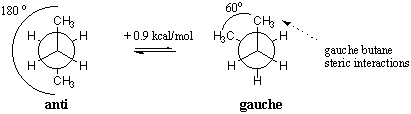 This effect is present in 1,2-difluoroethane (H 2 FCCFH 2) for which the gauche conformation is more stable by 2.4 to 3.4 kJ/mole in the gas phase.
This effect is present in 1,2-difluoroethane (H 2 FCCFH 2) for which the gauche conformation is more stable by 2.4 to 3.4 kJ/mole in the gas phase.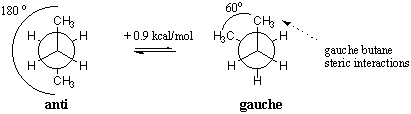 The fluorine gauche effect in 1,2‐difluoroethane and the related anomeric effect.
The fluorine gauche effect in 1,2‐difluoroethane and the related anomeric effect.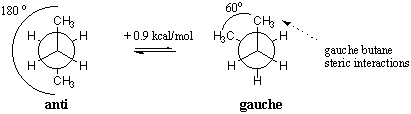 This effect is present in 1,2-difluoroethane (H 2 FCCFH 2) for which the gauche conformation is more stable by 2.4 to 3.4 kJ/mole in the gas phase.
This effect is present in 1,2-difluoroethane (H 2 FCCFH 2) for which the gauche conformation is more stable by 2.4 to 3.4 kJ/mole in the gas phase.